Over the past four decades, scientific interest in sperm DNA fragmentation (SDF) has increased substantially, establishing it as an important diagnostic biomarker of paternal genome integrity and male fertility potential. Advances in reproductive medicine have shown that conventional semen parameters do not always fully explain infertility, particularly in couples experiencing recurrent implantation failure, unexplained infertility, or repeated pregnancy loss. As a result, SDF has emerged as a clinically relevant marker that provides additional insight into sperm quality and reproductive outcomes (Pardiñas et al., 2022).
Sperm DNA fragmentation refers to breaks within the DNA strands of spermatozoa and is broadly classified into two main types: single-strand DNA fragmentation (ssSDF) and double-strand DNA fragmentation (dsSDF) (Agarwal et al., 2020). These two forms of DNA damage differ not only in their structure but also in their biological consequences. Single-strand breaks involve damage to only one of the DNA strands, whereas double-strand breaks affect both strands simultaneously, resulting in more severe genomic instability. Both forms can impair fertility, but dsSDF is considered particularly detrimental due to its limited capacity for repair after fertilization.
SDF arises from a complex interplay of internal and external factors that affect sperm development, maturation, and function. Internal factors include problems that arise during sperm development, such as improper DNA packaging, natural cell death processes that do not complete correctly, increased oxidative stress, aging, and inherited genetic traits. External factors include lifestyle and environmental influences such as smoking, excess weight, exposure to heat, air pollution, hormone-disrupting chemicals, infections, and certain medical conditions like diabetes and varicocele. Many of these internal and external factors increase oxidative stress, leading to an overproduction of reactive oxygen species (ROS), which is a major cause of sperm DNA damage.
The clinical implications of SDF vary depending on the type and extent of DNA damage. Elevated levels of ssSDF have been associated with reduced progressive sperm motility and lower rates of natural conception, likely due to impaired sperm function and reduced ability to reach and fertilize the oocyte. In contrast, dsSDF has a more pronounced negative impact on reproductive outcomes. High levels of dsSDF have been linked to compromised embryo development, reduced implantation rates, increased risk of early pregnancy loss, and higher rates of first-trimester miscarriage (Casanovas et al., 2019; Ribas-Maynou et al., 2012b; Ribas-Maynou & Benet, 2019).
Given the importance of sperm DNA integrity, considerable attention has been directed toward laboratory techniques that can reduce SDF and improve sperm selection for assistedreproductive technologies (ART). One such innovation is microfluidic sperm selection. Microfluidic technology is designed to replicate the conditions and natural selection mechanisms that occur within the female reproductive tract. Unlike conventional sperm preparation methods, microfluidics relies on sperm motility and natural movement rather than external mechanical forces.
Microfluidic sperm selection employs a gentle, non-invasive approach that avoids centrifugation, a process commonly used in traditional methods such as density gradient centrifugation (DGC) and swim-up. Centrifugation exposes sperm to high gravitational forces that may increase oxidative stress and contribute to DNA damage. In contrast, microfluidic systems allow sperm to migrate through precisely engineered microchannels under controlled fluid dynamics, favoring the selection of motile, morphologically normal, and functionally competent sperm. The primary goal of this approach is to minimize sperm DNA fragmentation while isolating sperm with superior fertilization potential.
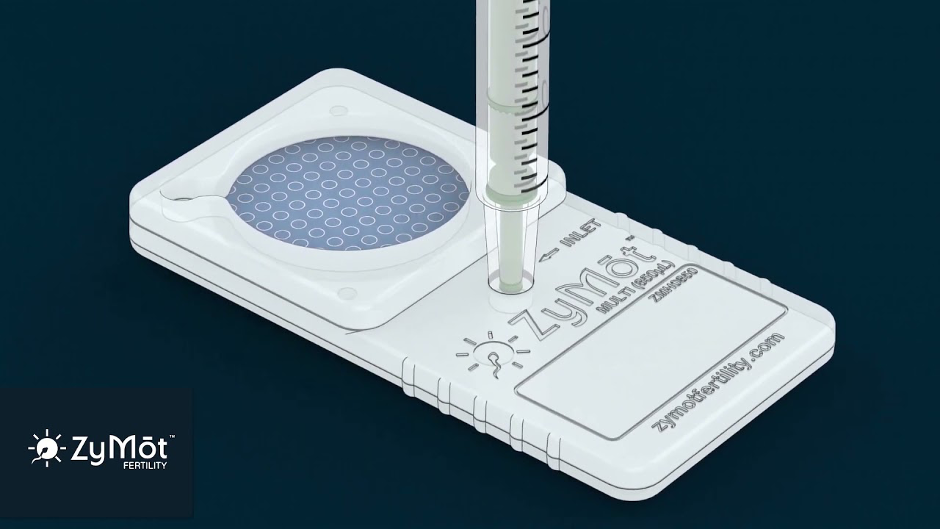
One of the most widely studied and clinically used microfluidic sperm-selection systems is the ZyMot platform, which applies microfluidic technology to mimic natural sperm selection and was developed based on earlier experimental research before being commercialized for clinical use (Pardiñas et al., 2022). For ICSI procedures, the ZyMot ICSI chip is used, as it is designed to isolate a small number of high-quality sperm suitable for direct injection into the egg. In contrast, for male fertility testing, the ZyMot Multi (850 µL) device is used, as a larger volume of selected sperm is required to allow for comprehensive laboratory analysis. Both devices operate on the same microfluidic principles but are tailored to different clinical and diagnostic needs.
The dimensions and design of the microfluidic channel are critical to its function. The channel creates hydrodynamic conditions that restrict the passage of sperm with poor motility or abnormal morphology, while allowing stronger, normally shaped sperm to progress. This concept was first described in earlier experimental studies (Zhang et al., 2011) and later validated in clinical settings (Quinn et al., 2018). As a result, the ZyMot device selectively enriches sperm populations with improved functional and genetic quality.
Multiple studies have demonstrated the benefits of ZyMot-selected sperm in terms of DNA integrity. Quinn et al. (2018) reported that sperm isolated using the ZyMot ICSI chip exhibited undetectable levels of sperm DNA fragmentation when assessed using the sperm chromatin dispersion (SCD) test. In contrast, measurable DNA fragmentation was observed in unprocessed semen samples and in samples prepared using conventional DGC followed by swim-up. These findings suggest that microfluidic selection may be more effective at minimizing DNA damage than traditional centrifugation-based techniques.
More recent evidence further supports these observations. Pujol et al. (2022) compared raw semen samples and swim-up–processed samples with sperm prepared using the commercially available ZyMot ICSI device. In men with initially high levels of sperm DNA fragmentation, defined as greater than 60%, microfluidic processing resulted in a 46% reduction in double-strand DNA fragmentation, as measured by the neutral Comet assay. This finding is particularly significant given the strong association between dsSDF and adverse reproductive outcomes.
In addition to the ZyMot device, other microfluidic platforms such as the SwimCount™ Harvester have been developed and evaluated. Comparative studies have shown that microfluidic devices consistently outperform traditional sperm preparation methods. When compared with density gradient centrifugation, the SwimCount™ Harvester demonstrated significantly improved outcomes in progressive sperm motility, total progressive motile sperm count, sperm vitality, normal morphology, and reduced sperm DNA fragmentation. Comparisons with the swim-up technique revealed statistically significant improvements in purified sperm concentration, total progressive motile sperm count, and vitality.
Beyond improvements in sperm quality, microfluidic technologies offer several practical advantages in the laboratory setting. These systems require smaller volumes of culture media, reduce dependence on complex laboratory equipment, and shorten processing times. The simplified workflow reduces overall laboratory workload, minimizes sample handling, and lowers the risk of human error. Together, these factors contribute to a more standardized and reproducible sperm selection process.
References:
Pardiñas, M. L., Martin, A., Ortega-Jaén, D., De los Santos, J. M., Viloria, T., Gamiz, P., & De los Santos, M. J. (2022). Sperm DNA fragmentation and microfluidics: A new era in human sperm selection. Medicina Reproductiva y Embriología Clínica, 9(3), 100121. https://doi.org/10.1016/j.medre.2022.100121
Agarwal, A., Majzoub, A., Baskaran, S., Panner Selvam, M. K., Cho, C. L., Henkel, R., Finelli, R., Leisegang, K., Sengupta, P., Barbarosie, C., Parekh, N., Alves, M. G., Ko, E., Arafa, M., Tadros, N., Ramasamy, R., Kavoussi, P., Ambar, R., Kuchakulla, M., ... Shah, R. (2020). Sperm DNA Fragmentation: A New Guideline for Clinicians. The World Journal of Men’s Health, 38(4), 412–471. https://doi.org/10.5534/wjmh.200128
Ribas-Maynou, J., García-Peiró, A., Fernandez-Encinas, A., Amengual, M. J., Prada, E., Cortés, P., Navarro, J., & Benet, J. (2012b). Double Stranded Sperm DNA Breaks, Measured by Comet Assay, Are Associated with Unexplained Recurrent Miscarriage in Couples without a Female Factor. PLoS ONE, 7(9), e44679. https://doi.org/10.1371/journal.pone.0044679
Casanovas, A., Ribas-Maynou, J., Lara-Cerrillo, S., Jimenez-Macedo, A. R., Hortal, O., Benet, J., Carrera, J., & García-Peiró, A. (2019a). Double-stranded sperm DNA damage is a cause of delay in embryo development and can impair implantation rates. Fertility and Sterility, 111(4), 699- 707.e1. https://doi.org/10.1016/j.fertnstert.2018.11.035
Ribas-Maynou, J., & Benet, J. (2019). Single and Double Strand Sperm DNA Damage: Different Reproductive Effects on Male Fertility. Genes, 10(2), 105. https://doi.org/10.3390/genes10020105
Zhang, X., Khimji, I., Gurkan, U. A., Safaee, H., Catalano, P. N., Keles, H. O., Kayaalp, E., & Demirci, U. (2011). Lensless imaging for simultaneous microfluidic sperm monitoring and sorting. Lab on a Chip, 11(15), 10.1039/c1lc20236g. https://doi.org/10.1039/c1lc20236g
Quinn, M. M., Jalalian, L., Ribeiro, S., Ona, K., Demirci, U., Cedars, M. I., & Rosen, M. P. (2018).
Microfluidic sorting selects sperm for clinical use with reduced DNA damage compared to density gradient centrifugation with swim-up in split semen samples. Human Reproduction, 33(8), 1388–1393. https://doi.org/10.1093/humrep/dey239